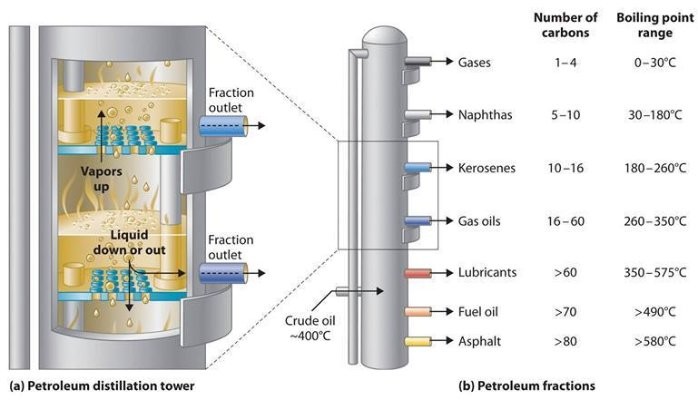
The problem with crude oil is that it contains hundreds of different types of hydrocarbons, all mixed together.We need to separate different kinds of hydrocarbon to extract useful substances.Fortunately, there’s an easy way to separate these substances, and that’s oil refining.
The oil refining process begins with a fractionating column.
As the length of the hydrocarbon chain increases, the boiling point of the hydrocarbon will gradually increase, so it can be separated completely by distillation.This is what happens in a refinery–at one stage of the refining process,that crude oil is heated, and at different evaporation temperatures, different lengths of hydrocarbon chains are separated.Each chain of different lengths has different properties and thus corresponds to different USES.
To understand the diversity of crude oil components and why oil refining is so important to society, let’s look at the products made from crude oil listed below:
Petroleum gas – used to heat, cook and make plastics
- small molecular alkane (1-4 carbon atoms)
- commonly known as methane, ethane, propane and butane
- boiling range = below 40 ℃
- are often pressurized liquefied for LPG (liquefied petroleum gas)
- naphtha or light oil, an intermediate product, will be further processed into petrol
A mixture of alkanes containing 5-9 carbon atoms
- boiling range = 60-100 ℃
- gasoline engine’s fuel
- liquid
- alkanes and naphthenes (5-12 carbon atoms)
- boiling range = 40-205 ℃
- kerosene, jet engine and tractor fuel;Raw materials for making other products
- liquid
- alkane (10-18 carbon atoms) and a mixture of aromatic hydrocarbon
- boiling range = 175-325 ℃
- diesel or fractional distillation diesel – used as diesel fuel or heating oil;Raw materials for making other products
- liquid
Carbon is greater than the equivalent of 12 alkanes
- boiling range = 250-350 ℃
- lubricating oil, used engine oil, grease and other lubricants
- liquid
- long chain (20-50 carbon atoms) alkane, cyclanes and aromatichydrocarbon
- boiling range = 300-370 ℃
- heavy oil or fuel oil, used as industrial fuel;Raw materials for making other products
- liquid
- long chain (20-70 carbon atoms) of alkanes, cyclanes and aromatic hydrocarbon
- boiling range = 370-600 ℃
- residual coke, asphalt, tar and wax;Raw materials for making other products
- solid
Carbon is greater than the equivalent of 70 polycyclic compounds
- boiling range = higher than 600 ℃
As you may have noticed, all of these products have different sizes and boiling ranges, and it is these properties that chemists use to refine petroleum