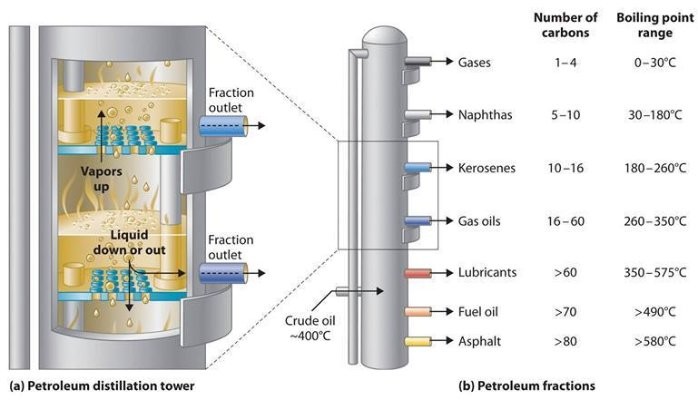
While the technique of refining crude oil is nowhere close to as complex as the method for harnessing nuclear vitality, it is still fairly ingenious and presents an inside look at considered one of our most controversial, important commodities. The first step in refining crude oil is known as fractional distillation.
We’ll have a look at the refining process in a broader view and then break down the steps concerned in fractional distillation. If you wish to see the larger image involved in oil, get a new perspective with this course on exploring the complexities of globalization and how it shapes the financial landscape.
Crude oil, also referred to as petroleum, is a mixture of a whole lot of helpful and worthless compounds. It is the refining refining process that not only separates these compounds, however even distinguishes different types and grades of priceless substances.
Hydrocarbons
An important substances in crude oil are hydrocarbons (because the identify suggests, they’re molecules made from hydrogen and carbon atoms). Hydrocarbons are particularly priceless as a result of they’re wealthy in vitality (clearly) and exist in diverse varieties. From methane to tar, there’s a hydrocarbon for every utility. Hydrocarbons are differentiated by their chain size, which refers to the variety of carbon atoms per molecule, which within the case of crude oil vary from 1 to 70+. The smaller the chain size, the decrease the boiling temperature, which is of the utmost significance. Gain a actually elemental understanding of these chemical properties with this superior on-line class on chemistry of the elements.
Chemicals And Blending
The first thing that happens to crude oil is that it is fractionally distilled, though trendy technology has precipitated a shift to chemical distillation, by using excessive-tech chemical processes and reactions to separate the completely different grades of hydrocarbons (though in some cases they need to combine hydrocarbons, a process often called unification). However I’ll save the remainder of the submit for the distillation course of. As soon as distillation is accomplished, the compounds are treated to eradicate impurities and then blended right into a remaining product, whether it be gasoline, jet gas or simple lubricants.
If you wish to learn about crude oil from a real chemical perspective, check out the third lecture in this high-rated Chemistry one hundred and one course, which deals with probably the most fascinating and related elements of chemistry: chemistry in society.
Fractional Distillation: The basics
As I discussed above, crude oil is a mixture of various compounds. These compounds all react differently; their chemical and even physical properties are different. These variations are precisely what permit us to distill oil, nonetheless there’s one distinction specifically that we’re interested by: boiling temperatures. Boiling temperatures are what enable fractional distillation to be each relatively easy and affordable. Even substances which have extremely comparable boiling temperatures can be accurately separated by fractional distillation.
A Step-By-Step Process
1. Heating
Naturally, to boil a substance, it’s essential to heat it. The completely different compounds in crude oil boil at extremely excessive temperatures, and due to the excessive quantities of potential vitality stored in these compounds, you will need to heat them consistently, safely and accurately. That is completed by way of high pressure steam, which is heated to over 1100 degrees Fahrenheit (660 C). At first, it will make sense to heat crude oil slowly, to allow the completely different compounds to boil at totally different temperatures. But it turns out that boiling temperatures solely come into effect after the oil is boiled.
2. Boiling
As soon as the crude oil boils, nearly all of the substances begin to turn to vapor, however some stay liquid. Substances that don’t boil at 1100F embody tar, asphalt, waxes, coke, etc. Substances will very low boiling points include gas (65F), gasoline (155F) and kerosene (200F), whereas issues like lubricant-stage oil and heavy gas boil above 600F.
Oil is a soiled enterprise in every sense of the term, but every source of vitality has its commerce-offs. White Home economic correspondent, Ben Ho, offers answers right here to questions surrounding vitality economics and the atmosphere.
Three. Fractional Columns
The substances that boil into vapor rise into a big, tall column (such as those seen in the picture above). That is the fractional distillation column, where all of the magic takes place. There are a series of accumulating plates which might be situated at numerous heights inside the column. The plates are designed to attract sure compounds, allowing these with decrease boiling temperatures (yes, lower, but extra on this soon) to ascend increased in the column. This maximizes the purity and quantity of substance collection.
But even high-grade, pure gasoline turns into a deadly pollutant when burned. For more data on oil disadvantages and different points with fashionable vitality, take a minute to read this informative submit on the varieties of environmental pollution and the best way to approach their causes.
Four. Rising . . . Cooling
As the vapor rises, it cools. The distillation column isn’t designed such that it sits directly above a boiling vat of oil. This wouldn’t present correct outcomes as the heat would rise and it will continue to heat the oil because it rose up the column. As a substitute, the crude oil is first fed into an enclosed steam boiler. Vapors are then transported to the distillation column through piping, thereby permitting the vapors to cool predictably.
5. Condensing
Like I stated beneath “Fractional Columns,substances with decrease boiling temperatures rise increased throughout the column. It’s because the vapors cool as they rise and condense, or turn into liquids. The substances that change between liquid and gas states at excessive temperatures will make this transition in a short time, turning again into liquids at the underside of the column. Different substances, corresponding to gasoline, will cool as they rise and subsequently attain a much larger peak. The connection between boiling point and column top is inversely associated: the decrease the boiling point, the upper within the column it is going to rise.
6. Collection
As planned, the gathering trays entice the liquids with unimaginable accuracy. Typically liquids that are still very hot can be transported to tanks for further cooling. Then it’s off to be blended, stored or chemically processed.
Oil reserves are depleting and well-liked favor is fading out. Still, our power options need quite a bit of work, as a lot of them nonetheless include appreciable disadvantages. Read this submit for 10 things you didn’t find out about wind power and why it’s not as clear as you think.
Life After Fractional Distillation
Chemical processing is starting to exchange fractional distillation as a result of it yields increased percentages of usable compounds per barrel of oil. As expected, this course of is barely more advanced than fractional distillation and that i don’t have time to enter it now. The treating and blending processes that comply with fractional distillation (or chemical distillation, for that matter), nevertheless, might be mentioned extra briefly.
Earlier than mixing can happen, distilled compounds are treated to eradicate impurities. These fluctuate from compound and compound, but the same old suspects are water, oxygen, nitrogen, sulfur and metals. Water is usually removed in a really previous-school course of, utilizing relatively simple strategies of absorption. Sulfur is handled extra chemically, usually by means of specifically designed sulfide compounds that acquire and bind to sulfur. Most of the remainder of the undesirables – nitrogen, oxygen, and so forth. – are passed via a sulfuric acid column, which speaks for itself.
 The final step is perhaps the only of all: rigorous mixing of pure compounds to make the final merchandise we buy, from gasoline to lubricants to jet gas. If you’re learning for Regents in chemistry are try to brush up on some basic information, try this 5-star, subject-by-matter assessment course on Regents Chemistry.
The final step is perhaps the only of all: rigorous mixing of pure compounds to make the final merchandise we buy, from gasoline to lubricants to jet gas. If you’re learning for Regents in chemistry are try to brush up on some basic information, try this 5-star, subject-by-matter assessment course on Regents Chemistry.
Here is more info about United visit our web site.